Difference between revisions of "Quantum mechanics"
(Undo revision 488588 by Lemonpeel - please respond to previous edit summary) |
(restore per talk) |
||
| Line 1: | Line 1: | ||
| − | '''Quantum mechanics''' | + | '''Quantum mechanics''' consists of the breakthrough in [[physics]] in the 1920s in understanding how particles behave inside [[atom]]s. Quantum mechanics posits that an [[electron]] (or any other [[sub-atomic particle]]) behaves as both a [[wave]] and a [[particle]]. |
| − | + | ||
| − | As a result of the wave nature of the electron, the position of the electron can never be precisely known. Whenever it is attempted to be measured, knowledge of the electron's | + | Classical mechanics, as initially discovered by [[Isaac Newton]], cannot explain atomic behavior. [[Erwin Schrodinger]] is generally credited with the formulation of the Schrodinger equation, around 1926. Other contributions were from [[Werner Heisenberg]], [[Niels Bohr]], [[John von Neumann]], and [[Hermann Weyl]]. |
| + | |||
| + | Classical mechanics would predict that an [[electron]] orbits a [[proton]] just as planets orbit the sun. Classical [[electromagnetism]] would predict that the orbiting electron would emit a time-varying electrical field just as a radio station does. But the electron would lose energy as it emits this [[radiation]], and would orbit closer and closer to the proton, until it collapses into the proton! Such a model cannot be correct. | ||
| + | |||
| + | As a result of the wave nature of the electron, the position of the electron can never be precisely known. Whenever it is attempted to be measured, knowledge of the electron's velocity is lost. Hence, there is an inherent uncertainty that prevents precisely measuring both the position and the momentum simultaneously. This is known as the [[Heisenberg Uncertainty Principle]]. | ||
| + | |||
| + | Quantum mechanics forms the basis for our understanding of chemical reactions, as well as all computers and electronic devices today. | ||
An important aspect of Quantum Mechanics is the predictions it makes about the [[radioactive decay]] of [[isotopes]]. Radioactive decay processes, controlled by the wave equations, are random events. A radioactive atom has a certain probability of decaying per unit time. As a result, the decay results in an exponential decrease in the amount of isotope remaining in a given sample as a function of time. The characteristic time required for 1/2 of the original amount of isotope to decay is known as the "half-life" and can vary from quadrillionths of a second (<sup>9</sup>B) to quintillions of years (<sup>186</sup>W). | An important aspect of Quantum Mechanics is the predictions it makes about the [[radioactive decay]] of [[isotopes]]. Radioactive decay processes, controlled by the wave equations, are random events. A radioactive atom has a certain probability of decaying per unit time. As a result, the decay results in an exponential decrease in the amount of isotope remaining in a given sample as a function of time. The characteristic time required for 1/2 of the original amount of isotope to decay is known as the "half-life" and can vary from quadrillionths of a second (<sup>9</sup>B) to quintillions of years (<sup>186</sup>W). | ||
| − | + | ==Key Idea== | |
| − | + | In quantum mechanics, we can no longer know what the outcome of an experiment will be. It only makes sense to ask what the ''probability'' of a particular outcome is. These probabilities are absolute squares of certain complex numbers called ''amplitudes'' associated to each possible outcome. | |
| − | + | ||
| + | Now the fundamental difference between quantum behavior and classical behavior is illustrated by the following example: Suppose that a particular outcome, A, of an experiment can happen in two ways, with amplitudes <math>z</math> and <math>w</math> (so that the corresponding probabilities are <math>||z||^2</math> and <math>||w||^2</math>. In classical physics, we would predict that the probability that A happens is just the sum of the probabilities of the two ways that A can happen--that is: <math>||z||^2+||w||^2</math>. Strangely, nature, for reasons unknown, does not appear to work this way. Instead, to get the probability for the outcome A, we have to add the amplitudes first, and only then square it. That is, the true probability for A is given by <math>||z+w||^2</math>. This means that the two amplitudes associated to the two possible ways A can happen can constructively or destructively interfere with each other. This gives rise the wave-like behavior of particles observed in situations such as the double-slit experiment. | ||
| + | |||
| + | ==Mathematics== | ||
| + | The mathematics of Quantum mechanics can be formulated in a number of ways: the "matrix mechanics" of Werner Heisenberg, the "path integrals" of [[Richard Feynman]], or the "wave mechanics" of Erwin Schrodinger. The most common formulation uses the language of infinite dimensional [[Hilbert Space]]s; observables such as position and momentum are [[operator]]s on such Hilbert Spaces. | ||
| + | ==Postulates of Quantum Mechanics== | ||
| + | {{jargon}} | ||
| + | *A physical state corresponds to a unit vector <math>\Psi</math> in a Hilbert space. | ||
| + | *Observable physical properties such as position, energy, and momentum are represented by self-adjoint operators on the Hilbert space. | ||
| + | *If we measure an observable <math>Q</math> of a physical state <math>\Psi</math>, the result of our measurement will be an eigenvalue <math>\lambda</math> for the operator <math>Q</math>. The probability that the measurement will yield the value <math>\lambda</math> is given by the norm-squared of the projection of <math>\Psi</math> onto the <math>\lambda</math>-eigenspace of <math>Q</math>. | ||
| + | *If we measure the observable <math>Q</math> and produce the value <math>\lambda</math>, the state <math>\Psi</math> collapses to its projection onto the <math>\lambda</math>-eigenspace of <math>Q</math>. | ||
| + | *A state <math>\Psi</math> evolves in time according the equation: <math>\Psi(t)=e^{iHt/\hbar}\Psi(0)</math>, where <math>H</math> is the energy operator. | ||
==External Links== | ==External Links== | ||
Revision as of 20:24, July 8, 2008
Quantum mechanics consists of the breakthrough in physics in the 1920s in understanding how particles behave inside atoms. Quantum mechanics posits that an electron (or any other sub-atomic particle) behaves as both a wave and a particle.
Classical mechanics, as initially discovered by Isaac Newton, cannot explain atomic behavior. Erwin Schrodinger is generally credited with the formulation of the Schrodinger equation, around 1926. Other contributions were from Werner Heisenberg, Niels Bohr, John von Neumann, and Hermann Weyl.
Classical mechanics would predict that an electron orbits a proton just as planets orbit the sun. Classical electromagnetism would predict that the orbiting electron would emit a time-varying electrical field just as a radio station does. But the electron would lose energy as it emits this radiation, and would orbit closer and closer to the proton, until it collapses into the proton! Such a model cannot be correct.
As a result of the wave nature of the electron, the position of the electron can never be precisely known. Whenever it is attempted to be measured, knowledge of the electron's velocity is lost. Hence, there is an inherent uncertainty that prevents precisely measuring both the position and the momentum simultaneously. This is known as the Heisenberg Uncertainty Principle.
Quantum mechanics forms the basis for our understanding of chemical reactions, as well as all computers and electronic devices today.
An important aspect of Quantum Mechanics is the predictions it makes about the radioactive decay of isotopes. Radioactive decay processes, controlled by the wave equations, are random events. A radioactive atom has a certain probability of decaying per unit time. As a result, the decay results in an exponential decrease in the amount of isotope remaining in a given sample as a function of time. The characteristic time required for 1/2 of the original amount of isotope to decay is known as the "half-life" and can vary from quadrillionths of a second (9B) to quintillions of years (186W).
Key Idea
In quantum mechanics, we can no longer know what the outcome of an experiment will be. It only makes sense to ask what the probability of a particular outcome is. These probabilities are absolute squares of certain complex numbers called amplitudes associated to each possible outcome.
Now the fundamental difference between quantum behavior and classical behavior is illustrated by the following example: Suppose that a particular outcome, A, of an experiment can happen in two ways, with amplitudes  and
and  (so that the corresponding probabilities are
(so that the corresponding probabilities are  and
and  . In classical physics, we would predict that the probability that A happens is just the sum of the probabilities of the two ways that A can happen--that is:
. In classical physics, we would predict that the probability that A happens is just the sum of the probabilities of the two ways that A can happen--that is:  . Strangely, nature, for reasons unknown, does not appear to work this way. Instead, to get the probability for the outcome A, we have to add the amplitudes first, and only then square it. That is, the true probability for A is given by
. Strangely, nature, for reasons unknown, does not appear to work this way. Instead, to get the probability for the outcome A, we have to add the amplitudes first, and only then square it. That is, the true probability for A is given by  . This means that the two amplitudes associated to the two possible ways A can happen can constructively or destructively interfere with each other. This gives rise the wave-like behavior of particles observed in situations such as the double-slit experiment.
. This means that the two amplitudes associated to the two possible ways A can happen can constructively or destructively interfere with each other. This gives rise the wave-like behavior of particles observed in situations such as the double-slit experiment.
Mathematics
The mathematics of Quantum mechanics can be formulated in a number of ways: the "matrix mechanics" of Werner Heisenberg, the "path integrals" of Richard Feynman, or the "wave mechanics" of Erwin Schrodinger. The most common formulation uses the language of infinite dimensional Hilbert Spaces; observables such as position and momentum are operators on such Hilbert Spaces.
Postulates of Quantum Mechanics
This article or section needs to be written in plain English, using plain English that most of our readers can understand. Articles that depend excessively on technical terms accessible only to specialists are useless for our purposes, so writers are admonished to avoid jargon
- A physical state corresponds to a unit vector
 in a Hilbert space.
in a Hilbert space. - Observable physical properties such as position, energy, and momentum are represented by self-adjoint operators on the Hilbert space.
- If we measure an observable
 of a physical state
of a physical state  , the result of our measurement will be an eigenvalue
, the result of our measurement will be an eigenvalue  for the operator
for the operator  . The probability that the measurement will yield the value
. The probability that the measurement will yield the value  is given by the norm-squared of the projection of
is given by the norm-squared of the projection of  onto the
onto the  -eigenspace of
-eigenspace of  .
. - If we measure the observable
 and produce the value
and produce the value  , the state
, the state  collapses to its projection onto the
collapses to its projection onto the  -eigenspace of
-eigenspace of  .
. - A state
 evolves in time according the equation:
evolves in time according the equation:  , where
, where 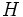 is the energy operator.
is the energy operator.
External Links
For an excellent discussion of quantum mechanics, see: http://www.chemistry.ohio-state.edu/betha/qm/
See also: Momentum (operator)