Difference between revisions of "Constants"
From Conservapedia
Bwilliston (Talk | contribs) (Fix typesetting of exponents in math mode) |
|||
| Line 11: | Line 11: | ||
== == | == == | ||
| − | Atomic mass unit: 1.00 amu = 1.67 x <math>10^-27</math>kg | + | Atomic mass unit: 1.00 amu = 1.67 x <math>10^{-27}</math>kg |
| − | Avagadro's number: 6.02 x <math>10^23</math> particles/mole | + | Avagadro's number: 6.02 x <math>10^{23}</math> particles/mole |
| − | Planck's constant: 6.23 x <math>10-34 \frac{Joules}{Hertz}</math> | + | Planck's constant: 6.23 x <math>10^{-34} \frac{Joules}{Hertz}</math> |
R = 0.0821 <math>\frac{1 \cdot atm}{mole \cdot K}</math> or 8.314 <math>\frac{dm^3 \cdot kPa}{mole \cdot K}</math> | R = 0.0821 <math>\frac{1 \cdot atm}{mole \cdot K}</math> or 8.314 <math>\frac{dm^3 \cdot kPa}{mole \cdot K}</math> | ||
Revision as of 03:47, March 10, 2007
1 atm = 101.1 kPa
1.000 calorie = 4.184 Joules
1 Calorie = 1000 calories
760 torr = 1 atm
760 mm Hg = 1 atm
Atomic mass unit: 1.00 amu = 1.67 x  kg
kg
Avagadro's number: 6.02 x 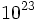 particles/mole
particles/mole
Planck's constant: 6.23 x 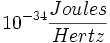
R = 0.0821  or 8.314
or 8.314 
Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm
| Mass | Charge | |
|---|---|---|
| Proton | 1.67 x  kg kg |
+1 |
| Neutron | 1.67 x  kg kg |
0 |
| Electron | 9.11 x 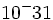 kg kg |
-1 |