Difference between revisions of "Constants"
From Conservapedia
(→Physical constants: added Faraday's) |
DavidB4-bot (Talk | contribs) (Spelling, grammar, and general cleanup, typos fixed: ie, → i.e.,, etc → etc.) |
||
| (4 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
| − | This page lists various constants used in [[physics]] and some useful conversions. | + | This page lists various '''constants''' used in [[physics]] and some useful conversions. |
==Physical constants== | ==Physical constants== | ||
The number in brackets represents the uncertainty in the constant, so that <math>6.626070040(81) \times 10^{-34}</math> has an uncertainty of <math>0.00000000081 \times 10^{-34}</math>. | The number in brackets represents the uncertainty in the constant, so that <math>6.626070040(81) \times 10^{-34}</math> has an uncertainty of <math>0.00000000081 \times 10^{-34}</math>. | ||
| − | {| class="wikitable" | + | {| class="wikitable" style="background: white" |
|- | |- | ||
!Name | !Name | ||
| Line 15: | Line 15: | ||
|[[Planck's constant]] | |[[Planck's constant]] | ||
|<math>h</math> | |<math>h</math> | ||
| − | |<math>6.626070040(81) \times 10^{-34} \,</math> | + | |<math>6.626070040(81) \times 10^{-34} \, \mathrm{J} \, \mathrm{s}</math> |
|It relates the [[frequency]] of a [[photon]] to its [[energy]] | |It relates the [[frequency]] of a [[photon]] to its [[energy]] | ||
|[[Quantum mechanics]] | |[[Quantum mechanics]] | ||
| Line 22: | Line 22: | ||
|[[Speed of light]] | |[[Speed of light]] | ||
|<math>c</math> | |<math>c</math> | ||
| − | |299,792,458 m s | + | |<math>299,792,458 \, \mathrm{m} \, \mathrm{s^{-1}}</math> |
|This is the speed of light in a [[vacuum]] | |This is the speed of light in a [[vacuum]] | ||
|Most of physics, such as [[optics]] and [[relativity]] | |Most of physics, such as [[optics]] and [[relativity]] | ||
| Line 29: | Line 29: | ||
|Permittivity of free space | |Permittivity of free space | ||
|<math>\epsilon_0</math> | |<math>\epsilon_0</math> | ||
| − | |<math>8.854187817... \times 10^{-12} | + | |<math>8.854187817... \times 10^{-12} \, \mathrm{F} \, \mathrm{m^{-1}}</math> |
|Related to the strength of an [[electric field]] produced by a charge | |Related to the strength of an [[electric field]] produced by a charge | ||
|[[Electromagnetism]] | |[[Electromagnetism]] | ||
| Line 36: | Line 36: | ||
|Permeability of free space | |Permeability of free space | ||
|<math>\mu_0</math> | |<math>\mu_0</math> | ||
| − | |<math>4 \pi \times 10^{-7}</math> | + | |<math>4 \pi \times 10^{-7} \, \mathrm{H} \, \mathrm{m^{-1}}</math> |
| − | |Related to the strength of [[magnetic field]s produced by a [[current]] | + | |Related to the strength of [[magnetic field]]s produced by a [[current]] |
|[[Electromagnetism]] | |[[Electromagnetism]] | ||
|Exact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?mu0</ref> | |Exact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?mu0</ref> | ||
| Line 43: | Line 43: | ||
|Elementary charge | |Elementary charge | ||
|<math>e</math> | |<math>e</math> | ||
| − | |<math>1.6021766208(98) \times 10^{-19}</math> | + | |<math>1.6021766208(98) \times 10^{-19} \, \mathrm{C}</math> |
|The magnitude of the electric charge on an electron | |The magnitude of the electric charge on an electron | ||
| − | |Determines the strength of electric and magnetic fields produced by [[proton]]s, [[electron]]s etc | + | |Determines the strength of electric and magnetic fields produced by [[proton]]s, [[electron]]s etc. |
|Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?e</ref> | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?e</ref> | ||
|- | |- | ||
|[[Avogadro's number]] | |[[Avogadro's number]] | ||
|<math>N_A</math> | |<math>N_A</math> | ||
| − | |<math>6.022140857(74) \times 10^{23} | + | |<math>6.022140857(74) \times 10^{23} \, \mathrm{mol^{-1}}</math> |
|The number of constituents in 1 mole of something | |The number of constituents in 1 mole of something | ||
|[[Chemistry]], [[statistical mechanics]], [[thermodynamics]] | |[[Chemistry]], [[statistical mechanics]], [[thermodynamics]] | ||
| Line 57: | Line 57: | ||
|Ideal gas constant | |Ideal gas constant | ||
|<math>R</math> | |<math>R</math> | ||
| − | |<math>8.3144598(48) | + | |<math>8.3144598(48) \, \mathrm{J} \, \mathrm{mol^{-1}} \, \mathrm{K^{-1}}</math> |
|Relates the [[pressure]], [[volume]], [[temperature]] and number of moles of an [[ideal gas]] | |Relates the [[pressure]], [[volume]], [[temperature]] and number of moles of an [[ideal gas]] | ||
|[[Thermodynamics]] | |[[Thermodynamics]] | ||
| Line 64: | Line 64: | ||
|Faraday Constant | |Faraday Constant | ||
|<math>F</math> | |<math>F</math> | ||
| − | |<math>96485 C | + | |<math>96485 \mathrm{C} \, \mathrm{mol^{-1}}</math> |
|The electric charge on one mole of electrons (Avogadro's number times elementary charge) | |The electric charge on one mole of electrons (Avogadro's number times elementary charge) | ||
|[[Chemistry]], [[physics]] | |[[Chemistry]], [[physics]] | ||
|Inexact<ref>http://ch302.cm.utexas.edu/echem/echem-stoich/echem-stoich-all.php</ref> | |Inexact<ref>http://ch302.cm.utexas.edu/echem/echem-stoich/echem-stoich-all.php</ref> | ||
| + | |- | ||
| + | |[[Gravitational constant]] | ||
| + | |<math>G</math> | ||
| + | |<math>6.67408(31) \times 10^{-11} \, \mathrm{N} \, \mathrm{m^2} \, \mathrm{kg^{-2}}</math> | ||
| + | |Constant related to the strength of Newtonian gravity | ||
| + | |Newtonian [[gravity]] | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?bg</ref> | ||
| + | |- | ||
| + | |Boltzmann constant | ||
| + | |<math>k</math> | ||
| + | |<math>1.38064852(79) \times 10^{-23} \, \mathrm{J} \, \mathrm{K^{-1}}</math> | ||
| + | |[[Thermodynamics]] | ||
| + | | | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?k</ref> | ||
| + | |- | ||
| + | |Reduced Planck's constant | ||
| + | |<math>\hbar</math> | ||
| + | |<math>1.054571800(13) \times 10^{-34} \, \mathrm{J} \, \mathrm{s}</math> | ||
| + | |[[Quantum mechanics]] | ||
| + | |It is [[Planck's constant]] divided by <math>2 \pi</math>. | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?hbar</ref> | ||
| + | |- | ||
| + | |Wien displacement constant | ||
| + | |<math>b</math> | ||
| + | |<math>2.8977729(17) \times 10^{-3} \, \mathrm{m} \, \mathrm{K}</math> | ||
| + | |Relates the [[temperature]] of a black body to the peak in its spectrum | ||
| + | | | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?bwien</ref> | ||
| + | |- | ||
| + | |[[Gravitational constant|Standard acceleration due to gravity]] | ||
| + | |<math>g</math> | ||
| + | |<math>9.80665 \, \mathrm{m} \, \mathrm{s^{-2}}</math> | ||
| + | |This is the [[acceleration]] of any object near to the earth's surface when [[gravity]] is the only force acting (i.e., no air resistance). | ||
| + | |[[Mechanics]] | ||
| + | |Exact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?gn</ref> | ||
| + | |- | ||
| + | |Proton mass | ||
| + | |<math>m_p</math> | ||
| + | |<math>1.672621898(21) \times 10^{-27} \, \mathrm{kg}</math> | ||
| + | |[[Rest mass]] of a [[proton]] | ||
| + | | | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?mp</ref> | ||
| + | |- | ||
| + | |Neutron mass | ||
| + | |<math>m_n</math> | ||
| + | |<math>1.674927471(21) \times 10^{-27} \, \mathrm{kg}</math> | ||
| + | |[[Rest mass]] of a [[neutron]] | ||
| + | | | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?mn</ref> | ||
| + | |- | ||
| + | |Electron mass | ||
| + | |<math>m_e</math> | ||
| + | |<math>9.10938356(11) \times 10^{-31} \, \mathrm{kg}</math> | ||
| + | |[[Rest mass]] of an [[electron]] | ||
| + | | | ||
| + | |Inexact<ref>http://physics.nist.gov/cgi-bin/cuu/Value?me</ref> | ||
|} | |} | ||
==Useful Conversions== | ==Useful Conversions== | ||
| − | + | ===Length=== | |
| − | 1. | + | 1 metre = 39.37 inches |
| + | 1 metre = 3.281 feet | ||
| + | 1 kilometre = 0.6213 miles | ||
| − | 1 | + | 1 mile = 5280 feet |
| + | 1 mile = 1760 yards | ||
| − | + | ===Weight=== | |
| − | + | 1 kilogram = 35.27 ounce | |
| + | 1 kilogram = 2.205 pounds | ||
| − | + | ===Pressure=== | |
| + | 1 atm = 101.1 kPa | ||
| + | 1 atm = 760 torr | ||
| + | 1 atm = 760 mm Hg | ||
| − | Atomic mass unit: 1.00 amu = 1.67 x < | + | ===Other=== |
| + | |||
| + | Atomic mass unit: 1.00 amu = 1.67 x 10<sup>−27</sup> kg | ||
| + | |||
| + | 1.000 calorie = 4.184 Joules | ||
Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm | Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm | ||
| − | ---- | + | ---- |
| + | |||
| + | Relative [[mass (science)|mass]] and [[charge]] of subatomic particles found in [[atom]]s. | ||
{| class="wikitable" style="text-align:left" | {| class="wikitable" style="text-align:left" | ||
| − | |+ | + | |+ Atomic particles |
|- | |- | ||
| − | ! !! Mass !! Charge | + | ! !! Relative Mass !! Relative Charge |
|- | |- | ||
| − | | '''Proton''' || 1 | + | | '''Proton''' || 1 || +1 |
|- | |- | ||
| − | | '''Neutron''' || 1 | + | | '''Neutron''' || 1 || 0 |
|- | |- | ||
| − | | '''Electron''' || | + | | '''Electron''' || 1/1837 || -1 |
|- | |- | ||
|} | |} | ||
| + | ==Prefixes== | ||
| + | Prefixes in front of a unit indicate that that unit should be multiplied by a multiple of ten, e.g. Giga is 10<sup>9</sup>, so 1 GPa = 10<sup>9</sup> Pa. | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | !Name | ||
| + | !Symbol | ||
| + | !Power | ||
| + | |- | ||
| + | |Pico | ||
| + | |p | ||
| + | |10<sup>−12</sup> | ||
| + | |- | ||
| + | |nano | ||
| + | |n | ||
| + | |10<sup>−9</sup> | ||
| + | |- | ||
| + | |micro | ||
| + | |μ | ||
| + | |10<sup>−6</sup> | ||
| + | |- | ||
| + | |milli | ||
| + | |m | ||
| + | |10<sup>−3</sup> | ||
| + | |- | ||
| + | |centi | ||
| + | |c | ||
| + | |10<sup>−2</sup> | ||
| + | |- | ||
| + | |deci | ||
| + | |d | ||
| + | |10<sup>−1</sup> | ||
| + | |- | ||
| + | |deca | ||
| + | |da | ||
| + | |10<sup>1</sup> | ||
| + | |- | ||
| + | |hecto | ||
| + | |h | ||
| + | |10<sup>2</sup> | ||
| + | |- | ||
| + | |kilo | ||
| + | |k | ||
| + | |10<sup>3</sup> | ||
| + | |- | ||
| + | |mega | ||
| + | |M | ||
| + | |10<sup>6</sup> | ||
| + | |- | ||
| + | |giga | ||
| + | |G | ||
| + | |10<sup>9</sup> | ||
| + | |- | ||
| + | |tera | ||
| + | |T | ||
| + | |10<sup>12</sup> | ||
| + | |} | ||
==References== | ==References== | ||
{{reflist}} | {{reflist}} | ||
Latest revision as of 02:00, July 17, 2019
This page lists various constants used in physics and some useful conversions.
Contents
Physical constants
The number in brackets represents the uncertainty in the constant, so that  has an uncertainty of
has an uncertainty of  .
.
| Name | Symbol | Value | Meaning | Used in | Exact |
|---|---|---|---|---|---|
| Planck's constant | 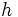
|

|
It relates the frequency of a photon to its energy | Quantum mechanics | Inexact[1] |
| Speed of light | 
|

|
This is the speed of light in a vacuum | Most of physics, such as optics and relativity | Exact[2] |
| Permittivity of free space | 
|

|
Related to the strength of an electric field produced by a charge | Electromagnetism | Exact[3] |
| Permeability of free space | 
|

|
Related to the strength of magnetic fields produced by a current | Electromagnetism | Exact[4] |
| Elementary charge | 
|

|
The magnitude of the electric charge on an electron | Determines the strength of electric and magnetic fields produced by protons, electrons etc. | Inexact[5] |
| Avogadro's number | 
|

|
The number of constituents in 1 mole of something | Chemistry, statistical mechanics, thermodynamics | Inexact[6] |
| Ideal gas constant | 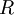
|

|
Relates the pressure, volume, temperature and number of moles of an ideal gas | Thermodynamics | Inexact[7] |
| Faraday Constant | 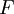
|

|
The electric charge on one mole of electrons (Avogadro's number times elementary charge) | Chemistry, physics | Inexact[8] |
| Gravitational constant | 
|

|
Constant related to the strength of Newtonian gravity | Newtonian gravity | Inexact[9] |
| Boltzmann constant | 
|

|
Thermodynamics | Inexact[10] | |
| Reduced Planck's constant | 
|

|
Quantum mechanics | It is Planck's constant divided by  . .
|
Inexact[11] |
| Wien displacement constant | 
|

|
Relates the temperature of a black body to the peak in its spectrum | Inexact[12] | |
| Standard acceleration due to gravity | 
|

|
This is the acceleration of any object near to the earth's surface when gravity is the only force acting (i.e., no air resistance). | Mechanics | Exact[13] |
| Proton mass | 
|

|
Rest mass of a proton | Inexact[14] | |
| Neutron mass | 
|

|
Rest mass of a neutron | Inexact[15] | |
| Electron mass | 
|

|
Rest mass of an electron | Inexact[16] |
Useful Conversions
Length
1 metre = 39.37 inches 1 metre = 3.281 feet 1 kilometre = 0.6213 miles
1 mile = 5280 feet 1 mile = 1760 yards
Weight
1 kilogram = 35.27 ounce 1 kilogram = 2.205 pounds
Pressure
1 atm = 101.1 kPa 1 atm = 760 torr 1 atm = 760 mm Hg
Other
Atomic mass unit: 1.00 amu = 1.67 x 10−27 kg
1.000 calorie = 4.184 Joules
Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm
Relative mass and charge of subatomic particles found in atoms.
| Relative Mass | Relative Charge | |
|---|---|---|
| Proton | 1 | +1 |
| Neutron | 1 | 0 |
| Electron | 1/1837 | -1 |
Prefixes
Prefixes in front of a unit indicate that that unit should be multiplied by a multiple of ten, e.g. Giga is 109, so 1 GPa = 109 Pa.
| Name | Symbol | Power |
|---|---|---|
| Pico | p | 10−12 |
| nano | n | 10−9 |
| micro | μ | 10−6 |
| milli | m | 10−3 |
| centi | c | 10−2 |
| deci | d | 10−1 |
| deca | da | 101 |
| hecto | h | 102 |
| kilo | k | 103 |
| mega | M | 106 |
| giga | G | 109 |
| tera | T | 1012 |
References
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?h
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?c
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?ep0
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?mu0
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?e
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?na
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?r
- ↑ http://ch302.cm.utexas.edu/echem/echem-stoich/echem-stoich-all.php
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?bg
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?k
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?hbar
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?bwien
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?gn
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?mp
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?mn
- ↑ http://physics.nist.gov/cgi-bin/cuu/Value?me