Difference between revisions of "Constants"
From Conservapedia
DavidB4-bot (Talk | contribs) (clean up & uniformity) |
|||
| Line 7: | Line 7: | ||
760 torr = 1 atm | 760 torr = 1 atm | ||
| − | 760 mm Hg = 1 atm | + | 760 mm Hg = 1 atm |
== == | == == | ||
| Line 36: | Line 36: | ||
|- | |- | ||
|} | |} | ||
| − | [[ | + | [[Category:Physics]] |
| − | [[ | + | [[Category:Chemistry]] |
Revision as of 06:49, July 13, 2016
1 atm = 101.1 kPa
1.000 calorie = 4.184 Joules
1 Calorie = 1000 calories
760 torr = 1 atm
760 mm Hg = 1 atm
Atomic mass unit: 1.00 amu = 1.67 x  kg
kg
Avogadro's number: 6.02 x 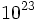 particles/mole
particles/mole
Planck's constant: 6.23 x 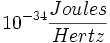
R = 0.0821  or 8.314
or 8.314 
Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm
| Mass | Charge | |
|---|---|---|
| Proton | 1.67 x  kg kg |
+1 |
| Neutron | 1.67 x  kg kg |
0 |
| Electron | 9.11 x 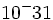 kg kg |
-1 |