Difference between revisions of "Constants"
From Conservapedia
(Replaced content with "REMEMBER ANON! WE SHALL LIVE ON UNTIL HIS DAY OF RECKONING!") |
|||
| Line 1: | Line 1: | ||
| − | + | 1 atm = 101.1 kPa | |
| + | |||
| + | 1.000 calorie = 4.184 Joules | ||
| + | |||
| + | 1 Calorie = 1000 calories | ||
| + | |||
| + | 760 torr = 1 atm | ||
| + | |||
| + | 760 mm Hg = 1 atm | ||
| + | |||
| + | == == | ||
| + | |||
| + | Atomic mass unit: 1.00 amu = 1.67 x <math>10^{-27}</math>kg | ||
| + | |||
| + | [[Avogadro's number]]: 6.02 x <math>10^{23}</math> particles/mole | ||
| + | |||
| + | Planck's constant: 6.23 x <math>10^{-34} \frac{Joules}{Hertz}</math> | ||
| + | |||
| + | R = 0.0821 <math>\frac{L \cdot atm}{mole \cdot K}</math> or 8.314 <math>\frac{J}{mole \cdot K}</math> | ||
| + | |||
| + | |||
| + | Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm | ||
| + | |||
| + | == == | ||
| + | |||
| + | {| class="wikitable" style="text-align:left" | ||
| + | |+ Bond Strengths | ||
| + | |- | ||
| + | ! !! Mass !! Charge | ||
| + | |- | ||
| + | | '''Proton''' || 1.67 x <math>10^-27 </math> kg || +1 | ||
| + | |- | ||
| + | | '''Neutron''' || 1.67 x <math>10^-27 </math> kg || 0 | ||
| + | |- | ||
| + | | '''Electron''' || 9.11 x <math>10^-31 </math> kg|| -1 | ||
| + | |- | ||
| + | |} | ||
| + | [[category:physics]] | ||
| + | [[category:chemistry]] | ||
Revision as of 14:44, August 21, 2011
1 atm = 101.1 kPa
1.000 calorie = 4.184 Joules
1 Calorie = 1000 calories
760 torr = 1 atm
760 mm Hg = 1 atm
Atomic mass unit: 1.00 amu = 1.67 x  kg
kg
Avogadro's number: 6.02 x 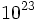 particles/mole
particles/mole
Planck's constant: 6.23 x 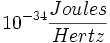
R = 0.0821  or 8.314
or 8.314 
Standard Temperature and Pressure (STP): temperature = 273 K and pressure = 1.00 atm
| Mass | Charge | |
|---|---|---|
| Proton | 1.67 x  kg kg |
+1 |
| Neutron | 1.67 x  kg kg |
0 |
| Electron | 9.11 x 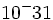 kg kg |
-1 |