Difference between revisions of "Periodic table of the elements"
m (Not in use anymore, I think) |
m (→The Periodic Table: replace with more traditional labeling scheme) |
||
| Line 21: | Line 21: | ||
!valign="bottom"|6A | !valign="bottom"|6A | ||
!valign="bottom"|7A | !valign="bottom"|7A | ||
| − | |{{ptc|2|He|Helium|4.003| | + | |{{ptc|2|He|Helium|4.003|gold}} |
|- | |- | ||
| − | |{{ptc|3|Li|Lithium|6.939}} | + | |{{ptc|3|Li|Lithium|6.939|pink}} |
| − | |{{ptc|4|Be|Beryllium|9.0122}} | + | |{{ptc|4|Be|Beryllium|9.0122|pink}} |
||||||||||||||||||| | ||||||||||||||||||| | ||
| − | |{{ptc|5|B|Boron|10.811}} | + | |{{ptc|5|B|Boron|10.811|#bbbbff}} |
| − | |{{ptc|6|C|Carbon|12.011}} | + | |{{ptc|6|C|Carbon|12.011|#00ff00}} |
|{{ptc|7|N|Nitrogen|14.007|#00ff00}} | |{{ptc|7|N|Nitrogen|14.007|#00ff00}} | ||
|{{ptc|8|O|Oxygen|15.999|#00ff00}} | |{{ptc|8|O|Oxygen|15.999|#00ff00}} | ||
|{{ptc|9|F|Fluorine|18.998|#00ff00}} | |{{ptc|9|F|Fluorine|18.998|#00ff00}} | ||
| − | |{{ptc|10|Ne|Neon|10.183| | + | |{{ptc|10|Ne|Neon|10.183|gold}} |
|- | |- | ||
| − | |{{ptc|11|Na|Sodium|22.99}} | + | |{{ptc|11|Na|Sodium|22.99|pink}} |
| − | |{{ptc|12|Mg|Magnesium|24.312}} | + | |{{ptc|12|Mg|Magnesium|24.312|pink}} |
!valign="bottom"|3B | !valign="bottom"|3B | ||
!valign="bottom"|4B | !valign="bottom"|4B | ||
| Line 45: | Line 45: | ||
!valign="bottom"|1B | !valign="bottom"|1B | ||
!valign="bottom"|2B | !valign="bottom"|2B | ||
| − | |{{ptc|13|Al|Aluminium|26.982}} | + | |{{ptc|13|Al|Aluminium|26.982|pink}} |
| − | |{{ptc|14|Si|Silicon|28.086}} | + | |{{ptc|14|Si|Silicon|28.086|#bbbbff}} |
| − | |{{ptc|15|P|Phosphorus|30.974}} | + | |{{ptc|15|P|Phosphorus|30.974|#00ff00}} |
| − | |{{ptc|16|S|Sulfur|32.064}} | + | |{{ptc|16|S|Sulfur|32.064|#00ff00}} |
|{{ptc|17|Cl|Chlorine|35.453|#00ff00}} | |{{ptc|17|Cl|Chlorine|35.453|#00ff00}} | ||
| − | |{{ptc|18|Ar|Argon|39.948| | + | |{{ptc|18|Ar|Argon|39.948|gold}} |
|- | |- | ||
| − | |{{ptc|19|K|Potassium|39.102}} | + | |{{ptc|19|K|Potassium|39.102|pink}} |
| − | |{{ptc|20|Ca|Calcium|40.08}} | + | |{{ptc|20|Ca|Calcium|40.08|pink}} |
| − | |{{ptc|21|Sc|Scandium|44.956}} | + | |{{ptc|21|Sc|Scandium|44.956|pink}} |
| − | |{{ptc|22|Ti|Titanium|47.9}} | + | |{{ptc|22|Ti|Titanium|47.9|pink}} |
| − | |{{ptc|23|V|Vanadium|50.942}} | + | |{{ptc|23|V|Vanadium|50.942|pink}} |
| − | |{{ptc|24|Cr|Chromium|51.996}} | + | |{{ptc|24|Cr|Chromium|51.996|pink}} |
| − | |{{ptc|25|Mn|Manganese|54.938}} | + | |{{ptc|25|Mn|Manganese|54.938|pink}} |
| − | |{{ptc|26|Fe|Iron|55.847}} | + | |{{ptc|26|Fe|Iron|55.847|pink}} |
| − | |{{ptc|27|Co|Cobalt|58.933}} | + | |{{ptc|27|Co|Cobalt|58.933|pink}} |
| − | |{{ptc|28|Ni|Nickel|58.71}} | + | |{{ptc|28|Ni|Nickel|58.71|pink}} |
| − | |{{ptc|29|Cu|Copper|63.546}} | + | |{{ptc|29|Cu|Copper|63.546|pink}} |
| − | |{{ptc|30|Zn|Zinc|65.37}} | + | |{{ptc|30|Zn|Zinc|65.37|pink}} |
| − | |{{ptc|31|Ga|Gallium|69.72| | + | |{{ptc|31|Ga|Gallium|69.72|pink}} |
| − | |{{ptc|32|Ge|Germanium|72.59}} | + | |{{ptc|32|Ge|Germanium|72.59|#bbbbff}} |
| − | |{{ptc|33|As|Arsenic|74.922}} | + | |{{ptc|33|As|Arsenic|74.922|#bbbbff}} |
| − | |{{ptc|34|Se|Selenium|78.96}} | + | |{{ptc|34|Se|Selenium|78.96|#00ff00}} |
| − | |{{ptc|35|Br|Bromine|79.904| | + | |{{ptc|35|Br|Bromine|79.904|#00ff00}} |
| − | |{{ptc|36|Kr|Krypton|83.8| | + | |{{ptc|36|Kr|Krypton|83.8|gold}} |
|- | |- | ||
| − | |{{ptc|37|Rb|Rubidium|85.47}} | + | |{{ptc|37|Rb|Rubidium|85.47|pink}} |
| − | |{{ptc|38|Sr|Strontium|87.62}} | + | |{{ptc|38|Sr|Strontium|87.62|pink}} |
| − | |{{ptc|39|Y|Yttrium|88.905}} | + | |{{ptc|39|Y|Yttrium|88.905|pink}} |
| − | |{{ptc|40|Zr|Zirconium|91.22}} | + | |{{ptc|40|Zr|Zirconium|91.22|pink}} |
| − | |{{ptc|41|Nb|Niobium|92.906}} | + | |{{ptc|41|Nb|Niobium|92.906|pink}} |
| − | |{{ptc|42|Mo|Molybdenum|95.94}} | + | |{{ptc|42|Mo|Molybdenum|95.94|pink}} |
| − | |{{ptc|43|Tc|Technetium|[97]| | + | |{{ptc|43|Tc|Technetium|[97]|pink}} |
| − | |{{ptc|44|Ru|Ruthenium|101.07}} | + | |{{ptc|44|Ru|Ruthenium|101.07|pink}} |
| − | |{{ptc|45|Rh|Rhodium|102.91}} | + | |{{ptc|45|Rh|Rhodium|102.91|pink}} |
| − | |{{ptc|46|Pd|Palladium|106.4}} | + | |{{ptc|46|Pd|Palladium|106.4|pink}} |
| − | |{{ptc|47|Ag|Silver|107.87}} | + | |{{ptc|47|Ag|Silver|107.87|pink}} |
| − | |{{ptc|48|Cd|Cadmium|112.4}} | + | |{{ptc|48|Cd|Cadmium|112.4|pink}} |
| − | |{{ptc|49|In|Indium|114.82}} | + | |{{ptc|49|In|Indium|114.82|pink}} |
| − | |{{ptc|50|Sn|Tin|118.69}} | + | |{{ptc|50|Sn|Tin|118.69|pink}} |
| − | |{{ptc|51|Sb|Antimony|121.75}} | + | |{{ptc|51|Sb|Antimony|121.75|#bbbbff}} |
| − | |{{ptc|52|Te|Tellurium|127.6}} | + | |{{ptc|52|Te|Tellurium|127.6|#bbbbff}} |
| − | |{{ptc|53|I|Iodine|126.9}} | + | |{{ptc|53|I|Iodine|126.9|#00ff00}} |
| − | |{{ptc|54|Xe|Xenon|131.3| | + | |{{ptc|54|Xe|Xenon|131.3|gold}} |
|- | |- | ||
| − | |{{ptc|55|Cs|Caesium|132.91}} | + | |{{ptc|55|Cs|Caesium|132.91|pink}} |
| − | |{{ptc|56|Ba|Barium|137.34}} | + | |{{ptc|56|Ba|Barium|137.34|pink}} |
| − | |{{ptc|57*|La|Lanthanum|138.91}} | + | |{{ptc|57*|La|Lanthanum|138.91|pink}} |
| − | |{{ptc|72|Hf|Hafnium|178.49}} | + | |{{ptc|72|Hf|Hafnium|178.49|pink}} |
| − | |{{ptc|73|Ta|Tantalum|180.95}} | + | |{{ptc|73|Ta|Tantalum|180.95|pink}} |
| − | |{{ptc|74|W|Tungsten|183.85}} | + | |{{ptc|74|W|Tungsten|183.85|pink}} |
| − | |{{ptc|75|Re|Rhenium|186.2}} | + | |{{ptc|75|Re|Rhenium|186.2|pink}} |
| − | |{{ptc|76|Os|Osmium|190.2}} | + | |{{ptc|76|Os|Osmium|190.2|pink}} |
| − | |{{ptc|77|Ir|Iridium|192.2}} | + | |{{ptc|77|Ir|Iridium|192.2|pink}} |
| − | |{{ptc|78|Pt|Platinum|195.09}} | + | |{{ptc|78|Pt|Platinum|195.09|pink}} |
| − | |{{ptc|79|Au|Gold|196.97}} | + | |{{ptc|79|Au|Gold|196.97|pink}} |
|{{ptc|80|Hg|Mercury (element)|200.59|pink}} | |{{ptc|80|Hg|Mercury (element)|200.59|pink}} | ||
| − | |{{ptc|81|Tl|Thallium|204.37}} | + | |{{ptc|81|Tl|Thallium|204.37|pink}} |
| − | |{{ptc|82|Pb|Lead|207.19}} | + | |{{ptc|82|Pb|Lead|207.19|pink}} |
| − | |{{ptc|83|Bi|Bismuth|208.98}} | + | |{{ptc|83|Bi|Bismuth|208.98|pink}} |
| − | |{{ptc|84|Po|Polonium|210}} | + | |{{ptc|84|Po|Polonium|210|pink}} |
| − | |{{ptc|85|At|Astatine|210}} | + | |{{ptc|85|At|Astatine|210|#bbbbff}} |
| − | |{{ptc|86|Rn|Radon|222| | + | |{{ptc|86|Rn|Radon|222|gold}} |
|- | |- | ||
| − | |{{ptc|87|Fr|Francium|215}} | + | |{{ptc|87|Fr|Francium|215|pink}} |
| − | |{{ptc|88|Ra|Radium|226.03}} | + | |{{ptc|88|Ra|Radium|226.03|pink}} |
| − | |{{ptc|89**|Ac|Actinium|227.03}} | + | |{{ptc|89**|Ac|Actinium|227.03|pink}} |
| − | |{{ptc|104|Rf|Rutherfordium|[261]| | + | |{{ptc|104|Rf|Rutherfordium|[261]|pink}} |
| − | |{{ptc|105|Db|Dubnium|[262]| | + | |{{ptc|105|Db|Dubnium|[262]|pink}} |
| − | |{{ptc|106|Sg|Seaborgium|[266]| | + | |{{ptc|106|Sg|Seaborgium|[266]|pink}} |
| − | |{{ptc|107|Bh|Bohrium|[264]| | + | |{{ptc|107|Bh|Bohrium|[264]|pink}} |
| − | |{{ptc|108|Hs|Hassium|[269]| | + | |{{ptc|108|Hs|Hassium|[269]|pink}} |
| − | |{{ptc|109|Mt|Meitnerium|[268]| | + | |{{ptc|109|Mt|Meitnerium|[268]|pink}} |
| − | |{{ptc|110|||[271]| | + | |{{ptc|110|||[271]|pink}} |
| − | |{{ptc|111|||[272]| | + | |{{ptc|111|||[272]|pink}} |
| − | |{{ptc|112|||[277]| | + | |{{ptc|112|||[277]|pink}} |
| | | | ||
| − | |{{ptc|114|||[289]| | + | |{{ptc|114|||[289]|pink}} |
| | | | ||
| − | |{{ptc|116|||[289]| | + | |{{ptc|116|||[289]|pink}} |
||| | ||| | ||
|- | |- | ||
| Line 130: | Line 130: | ||
|- | |- | ||
|colspan="2"|*Lanthanides | |colspan="2"|*Lanthanides | ||
| − | |{{ptc|58|Ce|Cerium|140.12}} | + | |{{ptc|58|Ce|Cerium|140.12|pink}} |
| − | |{{ptc|59|Pr|Praseodymium|140.91}} | + | |{{ptc|59|Pr|Praseodymium|140.91|pink}} |
| − | |{{ptc|60|Nd|Neodymium|144.24}} | + | |{{ptc|60|Nd|Neodymium|144.24|pink}} |
| − | |{{ptc|61|Pm|Promethium|145}} | + | |{{ptc|61|Pm|Promethium|145|pink}} |
| − | |{{ptc|62|Sm|Samarium|150.35}} | + | |{{ptc|62|Sm|Samarium|150.35|pink}} |
| − | |{{ptc|63|Eu|Europium|151.96}} | + | |{{ptc|63|Eu|Europium|151.96|pink}} |
| − | |{{ptc|64|Gd|Gadolinium|157.25}} | + | |{{ptc|64|Gd|Gadolinium|157.25|pink}} |
| − | |{{ptc|65|Tb|Terbium|158.92}} | + | |{{ptc|65|Tb|Terbium|158.92|pink}} |
| − | |{{ptc|66|Dy|Dysprosium|162.5}} | + | |{{ptc|66|Dy|Dysprosium|162.5|pink}} |
| − | |{{ptc|67|Ho|Holmium|164.93}} | + | |{{ptc|67|Ho|Holmium|164.93|pink}} |
| − | |{{ptc|68|Er|Erbium|167.26}} | + | |{{ptc|68|Er|Erbium|167.26|pink}} |
| − | |{{ptc|69|Tm|Thulium|168.93}} | + | |{{ptc|69|Tm|Thulium|168.93|pink}} |
| − | |{{ptc|70|Yb|Ytterbium|173.04}} | + | |{{ptc|70|Yb|Ytterbium|173.04|pink}} |
| − | |{{ptc|71|Lu|Lutetium|174.97}} | + | |{{ptc|71|Lu|Lutetium|174.97|pink}} |
||| | ||| | ||
|- | |- | ||
| Line 149: | Line 149: | ||
|- | |- | ||
|colspan="2"|**Actinides | |colspan="2"|**Actinides | ||
| − | |{{ptc|90|Th|Thorium|232.04}} | + | |{{ptc|90|Th|Thorium|232.04|pink}} |
| − | |{{ptc|91|Pa|Protactinium|231}} | + | |{{ptc|91|Pa|Protactinium|231|pink}} |
| − | |{{ptc|92|U|Uranium|238.03}} | + | |{{ptc|92|U|Uranium|238.03|pink}} |
| − | |{{ptc|93|Np|Neptunium|237.05}} | + | |{{ptc|93|Np|Neptunium|237.05|pink}} |
| − | |{{ptc|94|Pu|Plutonium|239.05}} | + | |{{ptc|94|Pu|Plutonium|239.05|pink}} |
| − | |{{ptc|95|Am|Americium|241.06}} | + | |{{ptc|95|Am|Americium|241.06|pink}} |
| − | |{{ptc|96|Cm|Curium|244.06}} | + | |{{ptc|96|Cm|Curium|244.06|pink}} |
| − | |{{ptc|97|Bk|Berkelium|249.08}} | + | |{{ptc|97|Bk|Berkelium|249.08|pink}} |
| − | |{{ptc|98|Cf|Californium|252.08}} | + | |{{ptc|98|Cf|Californium|252.08|pink}} |
| − | |{{ptc|99|Es|Einsteinium|252.08}} | + | |{{ptc|99|Es|Einsteinium|252.08|pink}} |
| − | |{{ptc|100|Fm|Fermium|257.1}} | + | |{{ptc|100|Fm|Fermium|257.1|pink}} |
| − | |{{ptc|101|Md|Mendelevium|258.1}} | + | |{{ptc|101|Md|Mendelevium|258.1|pink}} |
| − | |{{ptc|102|No|Nobelium|259.1}} | + | |{{ptc|102|No|Nobelium|259.1|pink}} |
| − | |{{ptc|103|Lr|Larencium|262.11}} | + | |{{ptc|103|Lr|Larencium|262.11|pink}} |
||| | ||| | ||
|- | |- | ||
| Line 168: | Line 168: | ||
|- | |- | ||
|style="background-color:#00ff00"| | |style="background-color:#00ff00"| | ||
| − | |colspan=17| | + | |colspan=17|[[Nonmetal]]s |
| + | |- | ||
| + | |style="background-color:#bbbbff"| | ||
| + | |colspan=17|[[Semimetal]]s | ||
|- | |- | ||
|style="background-color:pink"| | |style="background-color:pink"| | ||
| − | |colspan=17| | + | |colspan=17|[[Metal]]s |
| − | + | ||
| − | + | ||
| − | + | ||
|- | |- | ||
|style="background-color:gold"| | |style="background-color:gold"| | ||
| − | |colspan=17| | + | |colspan=17|[[Noble gas|Noble Gases]] |
| − | | | + | |
| − | + | ||
| − | + | ||
|} | |} | ||
[[Category:Chemistry]] | [[Category:Chemistry]] | ||
Revision as of 01:30, January 22, 2008
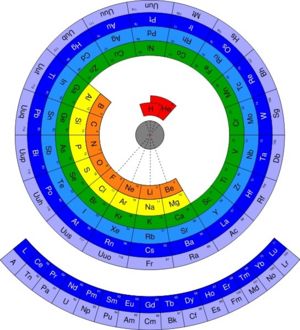
The periodic table of the elements is a diagrammatic tool to illustrate the scientific theory of periodicity in chemistry, in which the elements have similar properties based on their position on the table. For example, the elements on the left of the table are all metals. The table has a lot of predictive power, although there are many exceptions to the periodic properities.
The periodic table was first developed by Russian chemist Dimitri Mendeleev. Its layout accurately arranges elements by their electron configuration, atomic number, and chemical properties. Using the structure he created, Mendeleev predicted the existance and properties of several yet-undiscovered elements; when those elements were discovered, his estimates proved to be quite accurate.
The Periodic Table
| 1A | 8A | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2A | 3A | 4A | 5A | 6A | 7A | ||||||||||||
| 3B | 4B | 5B | 6B | 7B | <--- | 8B | --> | 1B | 2B | ||||||||
110
[271] |
111
[272] |
112
[277] |
114
[289] |
116
[289] |
|||||||||||||
| *Lanthanides | |||||||||||||||||
| **Actinides | |||||||||||||||||
| Nonmetals | |||||||||||||||||
| Semimetals | |||||||||||||||||
| Metals | |||||||||||||||||
| Noble Gases | |||||||||||||||||