Difference between revisions of "Periodic table of the elements"
m (add cat) |
MikeNookes (Talk | contribs) |
||
| Line 3: | Line 3: | ||
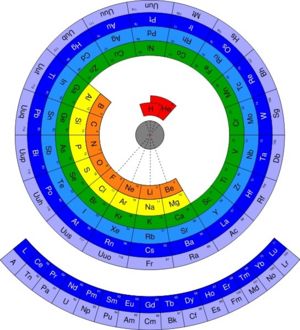
The '''periodic table of the elements''' is a diagrammatic tool to illustrate the scientific [[theory]] of [[periodicity]] in [[chemistry]], in which the [[element]]s have similar properties based on their position on the table. For example, the elements on the left of the table are all [[metal]]s. The table has a lot of predictive power, although there are many exceptions to the periodic properties. | The '''periodic table of the elements''' is a diagrammatic tool to illustrate the scientific [[theory]] of [[periodicity]] in [[chemistry]], in which the [[element]]s have similar properties based on their position on the table. For example, the elements on the left of the table are all [[metal]]s. The table has a lot of predictive power, although there are many exceptions to the periodic properties. | ||
| − | The periodic table was first developed by [[Russia]]n [[chemistry|chemist]] [[Dimitri Mendeleev]]. Its layout accurately arranges elements by their [[electron]] configuration, atomic number, and chemical properties. Using the structure he created, Mendeleev predicted the existence and properties of several yet-undiscovered elements; when those elements were discovered, his estimates proved to be quite accurate. | + | The periodic table was first developed by [[Russia]]n [[chemistry|chemist]] [[Dimitri Mendeleev]]. Its layout accurately arranges elements by their [[electron]] configuration, atomic number, and chemical properties. Using the structure he created, Mendeleev predicted the existence and properties of several yet-undiscovered elements; when those elements were discovered, his estimates proved to be quite accurate, vindicating his theory which had previously been criticised. |
{{clear}} | {{clear}} | ||
Revision as of 17:39, February 22, 2009
The periodic table of the elements is a diagrammatic tool to illustrate the scientific theory of periodicity in chemistry, in which the elements have similar properties based on their position on the table. For example, the elements on the left of the table are all metals. The table has a lot of predictive power, although there are many exceptions to the periodic properties.
The periodic table was first developed by Russian chemist Dimitri Mendeleev. Its layout accurately arranges elements by their electron configuration, atomic number, and chemical properties. Using the structure he created, Mendeleev predicted the existence and properties of several yet-undiscovered elements; when those elements were discovered, his estimates proved to be quite accurate, vindicating his theory which had previously been criticised.
External Links
- Interactive Periodic Table You've seen it before, but what do all those symbols and numbers really mean? You're about to find out.
The Periodic Table
| 1A | 8A | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2A | 3A | 4A | 5A | 6A | 7A | ||||||||||||
| 3B | 4B | 5B | 6B | 7B | <--- | 8B | --> | 1B | 2B | ||||||||
112
[277] |
114
[289] |
116
[289] |
|||||||||||||||
| *Lanthanides | |||||||||||||||||
| **Actinides | |||||||||||||||||
| Nonmetals | |||||||||||||||||
| Semimetals | |||||||||||||||||
| Metals | |||||||||||||||||
| Noble Gases | |||||||||||||||||